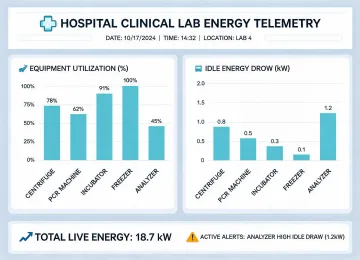
A 220-bed regional hospital ran aging chemistry and hematology instruments across two shifts. The facility team wanted to reduce electricity demand, but the laboratory could not accept a refresh plan that created downtime or forced unvalidated interface changes. Beckman Coulter began with a current-state review: analyzer age, idle draw, service events, reagent storage, sample routing, and the validation effort required to move any result-producing workflow.
"Sustainability is useful only when the night shift still has the instruments, reagents, and support needed to release patient results."
The practical roadmap combined scheduled replacement, service consolidation, operator retraining, and recycling certificates for retired equipment. The team prioritized analyzers with the highest idle draw and the weakest parts outlook, then built the change around planned validation windows instead of an arbitrary fiscal deadline. Energy savings were documented, but so were QC stability, downtime risk, and the training hours needed to keep operators comfortable after go-live.